| (2026.02) Shifting the surface oxidation cycle to a highvalence subsurface boosts water oxidation | |||||
| 작성자 | 관리자 | 작성일 | 2026-03-03 | 조회수 | 40 |
|---|---|---|---|---|---|
|
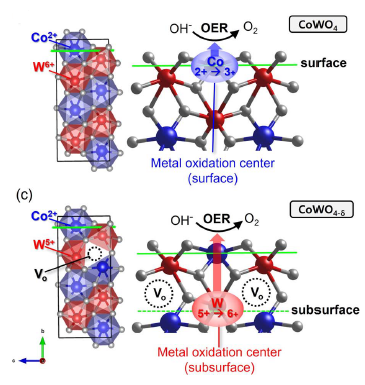
· 논문명 : Shifting the surface oxidation cycle to a highvalence subsurface boosts water oxidation · 저 자 : Chanseok Kim, Dasom Jeon, Jin Eun Heo, Hyeongoo Kim, Nayeong Kim, Seo Hyoung Chang*, Jungki Ryu*, Jun Hee Lee* · 게재지 : Journal of Materials Chemistry A (2026, 14, 6551-6559) · 초록 Molecular reactants' adsorption and subsequent redox reaction on a surface are two major processes in every electrochemical reaction. Thus, 3d-transition-metal (M) oxide has attracted significant attention because of its appropriate binding with the reactants and its facile metal oxidation cycle (Mn+ / M(n+1)+), which facilitates the reactants' redox reactions, such as oxygen evolution reaction (OER) (*OH / *O /*OOH / O2). However, the metal oxidation cycle on the surface still generates thermodynamic energy costs and causes surface degradation, thereby limiting efficient electrocatalytic processes. Here, we transfer the metal oxidation cycle from the surface to the non-catalytic subsurface. In a multivalent metal oxide CoWO4−d as a first showcase, we reveal a dramatic shift of the metal oxidation cycle from the Co(Co2+/Co3+) surface to an embedded but easily oxidizable W(W5+/W6+) subsurface. By moving the cycle to the stable subsurface, we relieve the repetitive surface metal oxidation during the OER and exploit the strong oxidation capability of the high-valence elements (W5+) protected in the subsurface. Disentangling the adsorption site and the oxidation cycle site is a powerful emerging strategy in electrocatalysis. This concept aligns with the paradigm of leveraging subsurface sites to overcome traditional scaling relations. Herein, we provide a clear demonstration of this principle in a multivalent CoWO4−d system, offering unprecedented synergistic effects, such as lowered overpotential and alleviated surface degradation, that can be generally applied to various electrochemical processes.
|
|||||